intro
Before going off to college to study physics, I promised my parents I'd come home knowing why magnets are magnetic. I realize now that I have two choices:
1. Never come home (because no one knows)
2. Come home and say just cuz.
Inspired by the best just cuz ever (delivered by Feynman), I've decided to choose the second option. However, I can do slightly better if i trade out answering why magnets work for answering how magnets work.
Working in Professor Eckert’s magnetism lab at Harvey Mudd College, I’m finally tackling the second question. This is the first of three articles on how magnets work. The three posts are:
1. Building Blocks
2. Domains and Magnetization
3. Application and Experiment
In this post, we will build magnetism from a microscopic level and explore different types of magnetism.
pt. 1 : building blocks
magnetism from electricity
If you stopped to watch Feynman, then you have been reminded of what happens when you play the Why? game. In order to avoid an infinite series of But why? we have to accept some baseline knowns.
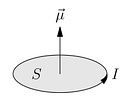
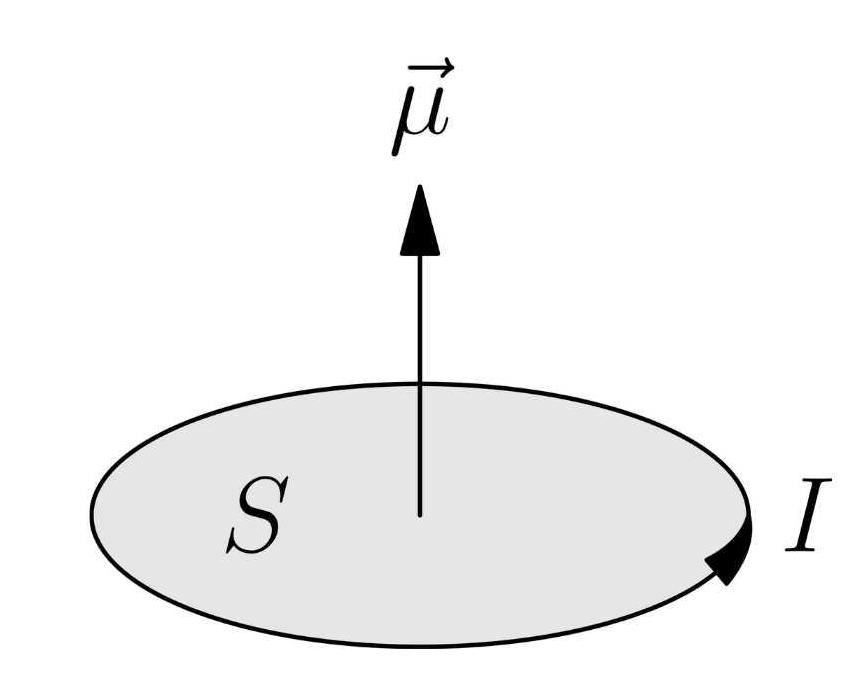
For this exploration, we will start with the fact that magnetism exists. More specifically, we will assert that an electric current generates a magnetic field. For some, this will be a fundamental assertion that’s easy to swallow. For others, this is a massive step that triggers the But why? response. Qualms aside, we will use the fact that a circular current results in a net magnetic moment pointing through the circle:
building up from electrons
The wire in this model has two traits:
It carries current (moving charges)
These moving charges follow a circle
An electron orbiting an atom is similar:
It carries current because it is charged
It has angular momentum from its atomic orbit as well as its spin. In other words, the electron is “moving in circles”
Therefore, like circular current-carrying wires, electrons have a magnetic moment vector.
Note: Electrons “orbiting” an atom is an incorrect classical model. However, it turns out that quantum mechanics yields an equivalent magnetic moment except for an extra multiplicative factor.
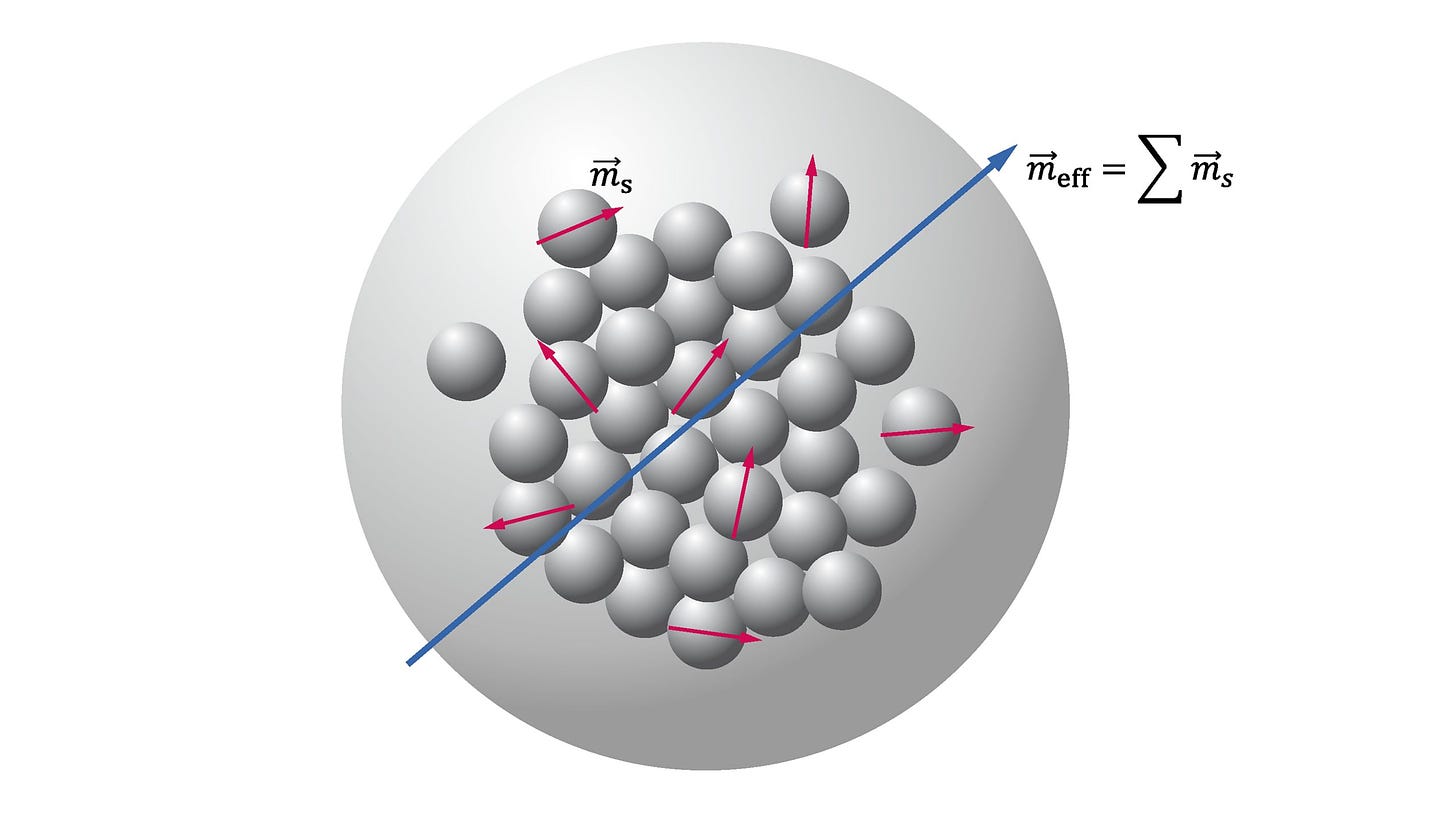
Thus, we can build a model of magnetism based off of each "orbiting" electron contributing a small vector magnetic moment. Electrons often pair up in orbitals, adopting opposite spin orientations and cancelling each other’s moments. However, in other materials electrons may be free-floating or unpaired. In this case, an atom or molecule may have a net moment. The alignment of these net moments lead to the macroscopic magnetic properties of a material.
types of magnetism
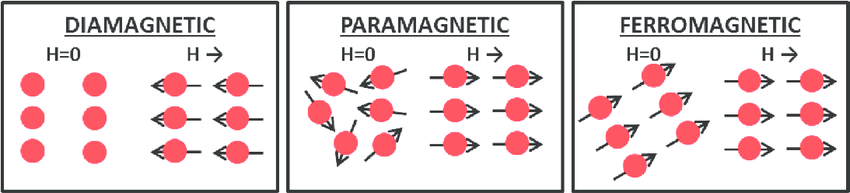
diamagnetism
Though magnetism feels like an elusive property, most all materials have a type of magnetism called diamagnetism. To understand this, we must take a second foray into electromagnetism to retrieve Lenz's Law.
Lenz's Law applies to a wire in a changing magnetic field. As the magnetic field through a loop of wire changes, the current in the loop adjusts to induce an opposing magnetic field.
Remembering that we can model electrons as loops of wire, Lenz’s law predicts that an increase in the applied magnetic field will induce electrons to generate an opposing magnetic field. The overall effect is for a diamagnetic material to be repelled in an applied magnetic field, a response known as diamagnetism.
paramagnetism
Diamagnetism is a weak effect, so if a material has another form of magnetism, it will dominate. Paramagnetism occurs when there are unpaired electrons, yielding atoms with a net magnetic moment. We can now envision all of the atoms in the material with a small moment vector. Importantly, in paramagnetic materials, these moments do not interact with one another, and so atomic perturbations like temperature cause these vectors to point randomly. Thus, at room temperature, paramagnets do not have a net magnetic moment.
However, in the presence of an external magnetic field, some of these moments align, and the material acquires a net moment. Due to this induced magnetization, paramagnets are attracted in a magnetic field (in contrast to the repulsion of diamagnets).
ferromagnetism
Ferromagnets are the classic magnets. Like paramagnets, the atoms in the material have net magnetic moments, but unlike paramagnets, the moments have strong interactions with one another. More specifically, the moments align and point the same direction as their neighbors. This results in a net magnetic moment, even in the absence of an applied field. Temperature still has a randomizing effect on ferromagnets, and if the temperature is high enough, the moments will be ripped from alignment, and a ferromagnet will become a paramagnet (at a point known as the Curie Temperature).
ferrimagnetism/antiferromagnetism
There are materials that have strongly interacting net moments at the atomic level but aren't ferromagnets.
First are the antiferromagnets. In these materials, moments are arranged to point in the opposite direction of their neighbor. This leads to an up-down-up-down arrangement of moments that results in a net 0 magnetic moment.
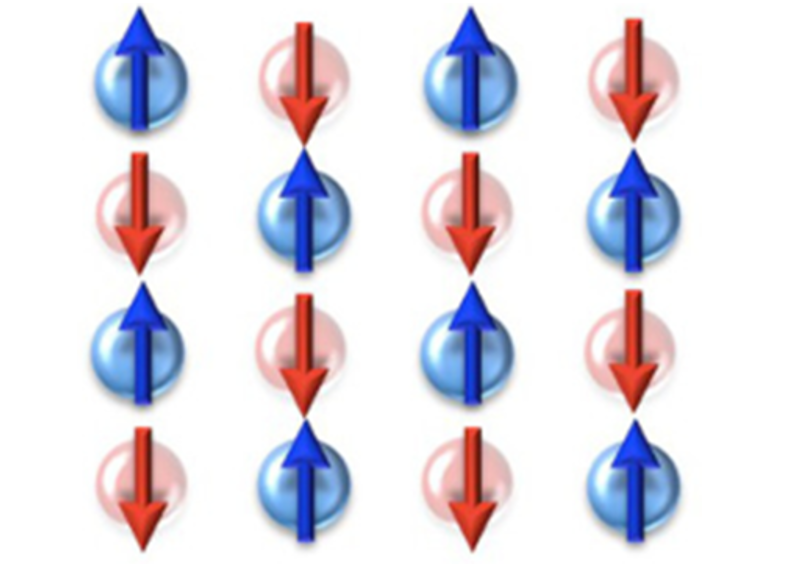
Next are the ferrimagnets (note the i instead of the o). Instead of having a single pattern of aligned moments, these materials have two patterns overlaid on one another. For example, one pattern could be an antiferromagnetic pattern (up, down, up, down) and another could be an “up, up, down” pattern. In this example, these patterns would combine to form a net moment in the up direction, since there are overall more “up” moments. Thus, ferrimagnets act similar to ferromagnets but with less magnetization.

summary
We've established an intuition for electron moments and described some categories of magnetism. Next post will explore domain theory and magnetic responses to changing magnetic fields.
part 2:
cited
Bruce M. Moskowitz's The Hitchiker's Guide to Magnetism
Lorenzo Calvano's undergraduate senior thesis Magnetic and Structural Properties of MnRh Thin Films.



Nice intro to magnetism! Could you talk more about how magnetization is measured in future articles?